|
You have signed an examinee agreement, and it will be enforced on this subreddit.ĭo not intentionally advertise paid or free products or services of any sort. We have one "stickied" post for each exam and score release day, contain all test day discussion/reactions to that thread only.ĭo not discuss any specific information from your actual MCAT exam. Importantly, methane lacks any lone pairs. For an example format for submitting pictures of questions from practice material click hereĭo not link to content that infringes on copyright laws (MCAT torrents, third party resources, etc).ĭo not post repeat "GOOD LUCK", "TEST SCORE", or test reaction posts. We consider both lone electron pairs and bond electron pairs while determining the shape of a molecule in electron geometry. These are considered spoilers and should be marked as such. Be nice to each other, hating on other users won't help you get extra points on the MCAT, so why do it?ĭo not post any question information from any resource in the title of your post. Rudeness or trolling will not be tolerated. Please message the moderators with your skills/ideas! MCAT RESOURCES & INFO Study Groups D With two nuclei around the central atom and one lone pair of electrons, the molecular geometry of SnCl 2 is bent, like SO 2, but with a ClSnCl bond angle of 95°. Want to help us improve this subreddit or tell us about a new resource we can add to the sidebar? Below you will find our forum rules, resources, and more. 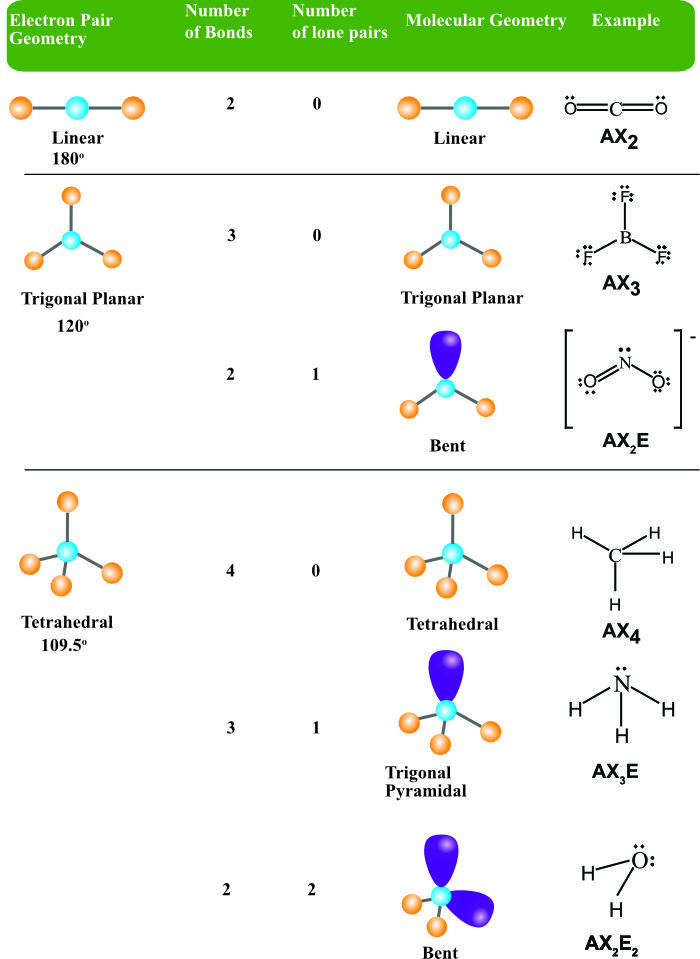
We request that you read the sidebar COMPLETELY before you post. r/MCAT is a place for support, discussion, advice, social networking, news, study tips and more. The MCAT (Medical College Admission Test) is offered by the AAMC and is a required exam for admission to medical schools in the USA and Canada. The oxygen has two bonding electron pairs (single bond to each H) and two non-bonding pairs giving water a #AX_2E_2# conformation and a bent shape.Welcome to the BEST place for MCAT prep and practice materials. #H_2O# we need to consider the central atom of water which is oxygen. We can use the following notations when examining a Lewis structure of a molecule.Į = non-bonding electron pairs of the central atom The molecular structure of the methane molecule, CH4 CH 4, is shown with a tetrahedral arrangement of the hydrogen atoms. This theory basically says that bonding and non-bonding electron pairs of the central atom in a molecule will repel (push away from) each other in three dimensional space and this gives the molecules their shape. VESPR stands for valence shell electron pair repulsion. 
It applies a theory called VESPR for short. Molecular geometry is a way of describing the shapes of molecules. For example, the shape of a water molecule (H2O) is bent, which gives it a high surface tension and allows it to dissolve many other substances. Similar logic applies to all the shapes, you just have to remember which "spoke" will be taken up by an electron pair. Thus, the electron-pair geometry is tetrahedral and the molecular structure is bent with an angle slightly less than 109.5°. Once there are any electron pairs, one spoke of the original shape gets "eaten up": for example, a #AX_4E_2# is an octahedron shape, but the two "spokes" are taken up by electron pairs, so you're left with just the square-a square planar shape. The Lewis structure of H 2 O indicates that there are four regions of high electron density around the oxygen atom: two lone pairs and two chemical bonds: Figure 7.6.9 7.6. #6#: octahedron (a flat square with two "spokes") #5#: trigonal bipyramid (a trigonal planar shape with two "spokes") 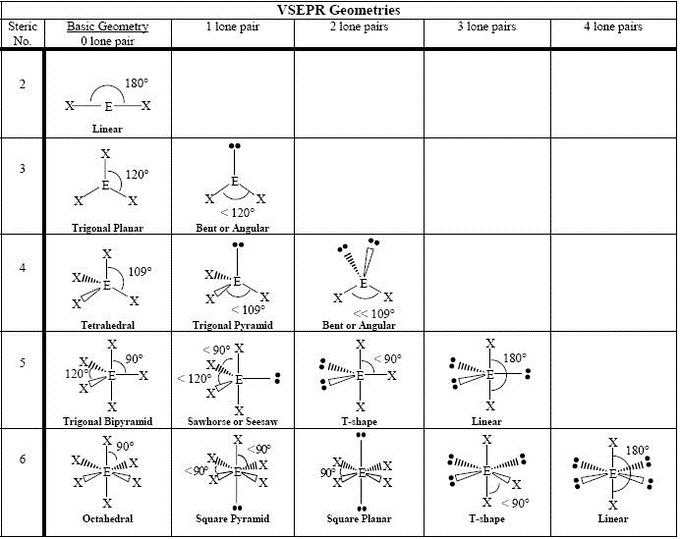
#3#: trigonal plane (a flat equilateral-triangle-looking shape) As it has a VSEPR shape #AX_5E_0# it is a trigonal bipyramid.Įach steric number has a same "basic shape": Examples of molecules with linear geometry are carbon dioxide (CO 2 ), beryllium chloride (BeCl 2 ), and nitric oxide (NO). The atoms are arranged in a straight line, and the angle between the bonds, or bond angle, is 180 °. Its steric number is #5# due to the #5# bonded atoms to the central #S# atom plus #0# lone electron pairs. Linear: It specifies the geometry shaped by a central atom surrounded by two other atoms. Thus, it is in the form #AX_3E_1#, which forms a trigonal pyramidal shape. You also treat double and triple bonds as one group instead of 3 just like you did in molecular shape. 
However, with electron geometry (steric number) lone pairs and bonds are treated the same. #N#, the central atom, has a steric number of #4#, calculated by the #3# atoms it's bonding with #+1# lone pair. In molecular shape (molecular geometry) you treat the electron’s lone pairs and the bonds as two different and separate things. This is the total number of electron pairs and bonds with other atoms. Find the central molecules' steric numbers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
